DataHub
In modern clinical trials, data from various sources—study sponsors, CROs, laboratories—and a wide range of analytical systems all converge. What remains are Excel spreadsheets, data silos, and manual processes that hinder collaboration, impede reproducibility, and delay results.
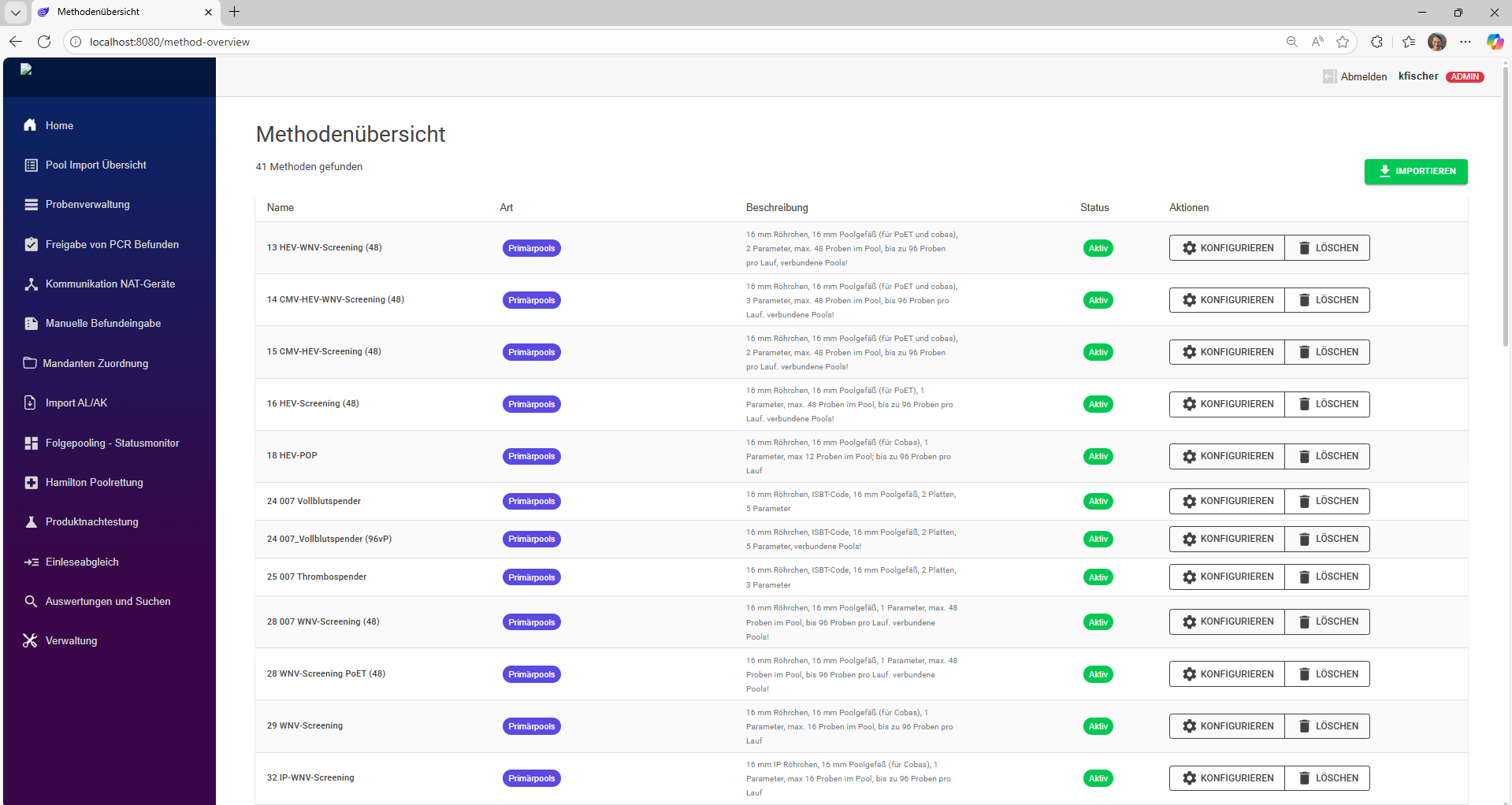
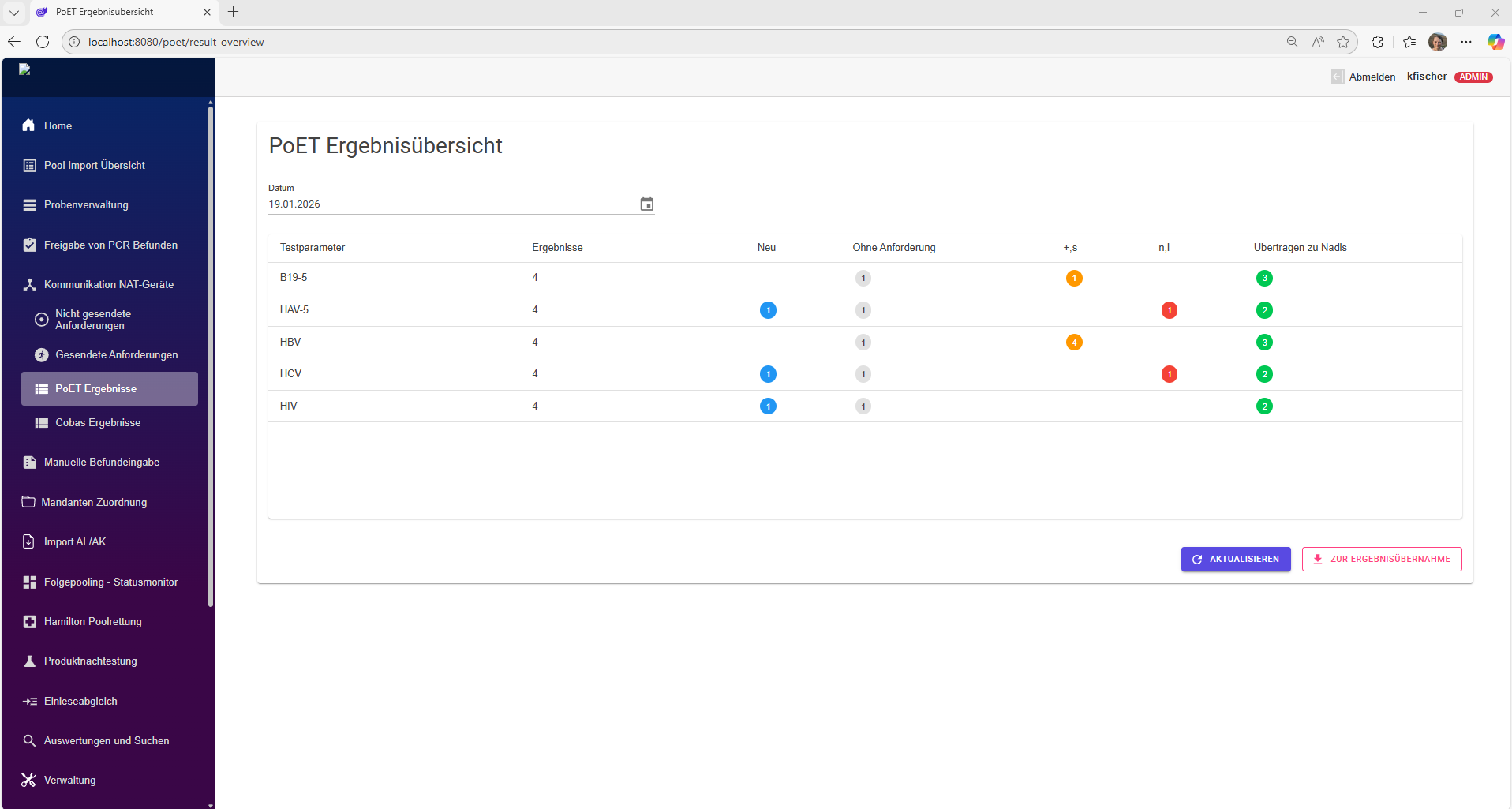
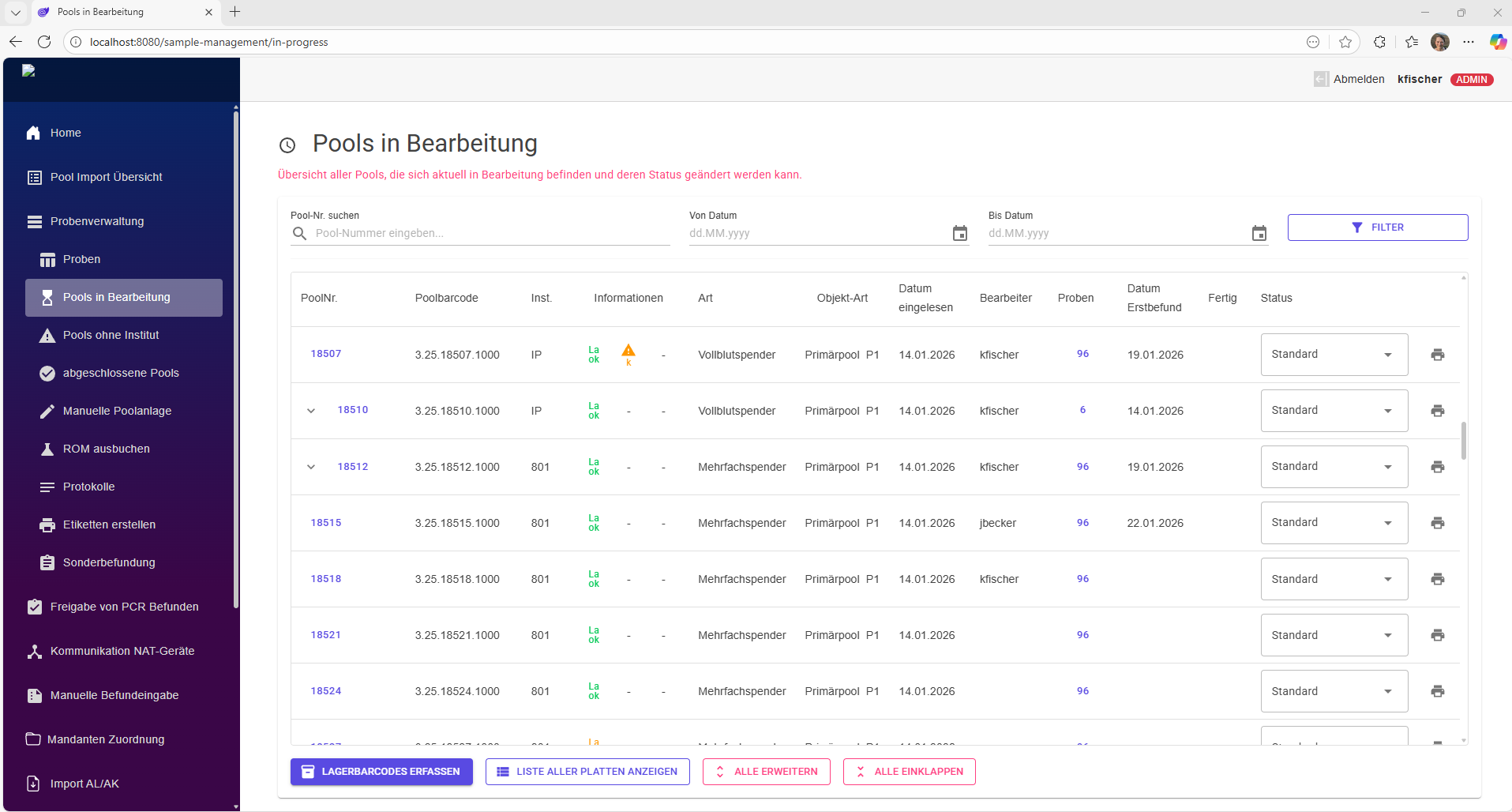
As a central platform, DataHub brings order to your clinical trial data.
Consistent data from the start to the end of the study
Modern studies examine numerous parameters that are collected by various specialized laboratories. This requires close cooperation between sponsors, contractors, CROs, and analytical laboratories. The resulting data is available in various formats and must be consolidated, stored in a structured manner, and evaluated—all in a way that is traceable and auditable at all times.
In practice, however, Excel spreadsheets, proprietary formats, and exported text files dominate, which are often combined manually or only partially automated. This approach is time-consuming, error-prone, and barely scalable.
DataHub puts an end to this Excel chaos.
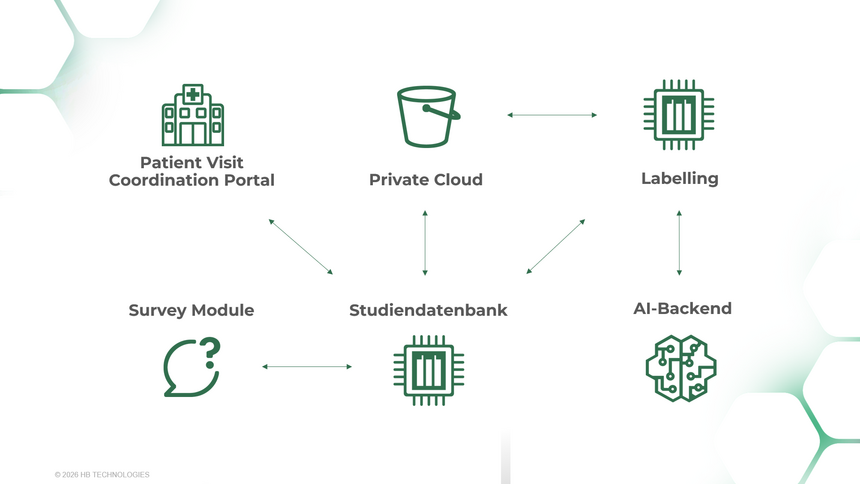
As a central platform for study data, DataHub enables smooth collaboration among all parties involved – from the beginning of the study to its completion. Data collection is structured and uniform, regardless of source or format. Changes during the course of the study, e.g., in evaluation criteria or evaluation logic, can be flexibly mapped and are documented transparently with versioning.
This creates a reliable, consistent database as the basis for robust study results. DataHub offers comprehensive functions for analysis and visualization – from classic statistical methods to AI-supported evaluation of complex data patterns.
Versatile Functions & High Expandability
DataHub provides you with the most essential features for the structured collection and evaluation of study data. The platform can be flexibly adapted to your studies.
- Study Database – Central management of studies, study participants, and users
- Patient Visit Coordination Portal – Management of subjects, multicenter studies, and subject appointments
- Individual Configuration of Study Structure – sites, subjects, visits, laboratory, etc.
- Survey Module – creation and evaluation of medical history forms and study-related questionnaires
- Secure & Independently Hosted – simple on-premise operation with Docker installation, full control over sensitive data, role-based user management
- Traceability & Audit Trail – Every analysis step documented and versioned, ideal for non-linear processes such as adjustment of data selection limits (gating) in flow cytometry
- Analysis & Visualization – Integrated statistical evaluations and visualizations
- AI-Supported Analytics – Data analytics based on voice input, reproducible analysis scripts that can be refined manually
- Human & Machine Labeling – Manual and AI-supported annotations for images, videos, and time series
- Open & Expandable – Modular architecture on an open source basis – flexibly adaptable to new studies, data formats, and analysis methods

Do you have questions regarding DataHub?
Book a free consultation or a live demo.
Congress Center (Basel, Schweiz)
Future Labs Live in Basel 2026
Meet Dr. Oliver Deusch, Chief Business Development Officer, and Dr. Marcus Rothe, Head of the Laboratory Equipment Division.
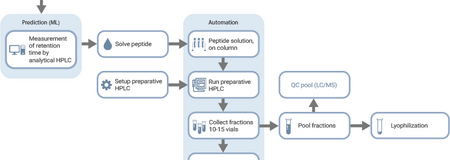
Machine Learning Accelerates Peptide Purification
Machine learning can now automate and accelerate the peptide purification process.
HB Technologies AG becomes distributor for OligoMaker oligonucleotide synthesizers in Europe
HB Technologies AG Becomes a Distributor for OligoMaker Oligonucleotide Synthesizers in Europe